 Hint: Count the number of atoms of each element, and then multiply that number by the element's atomic weight. Use the table below to find the atomic weight of each atom (element), or refer to a Periodic Table of the Elements. Let us calculate the molecular weight of some common compounds. For example, in one mole of a chemical compound there are 6.022 x 1023 molecules. One mole of 'something' contains 6.022 x 1023 entities. In both cases, it is the mass of 6.02 × 1023 molecules. One mole of carbon dioxide molecules has a mass of 44.01g. 1 dozen 12 items similarly we use the mole to. Just as we take a standard value to calculate different things e.g. A mole of any substance is 6.022×1023 molecules. atoms, molecules, ions, is defined as a mole. In a substance, the amount of entities present e.g. Explore molarity definition, uses, and applications in chemistry at BYJUS. The molar mass of any compound is the mass in grams of one mole of that compound. Molar mass of a substance is the mass in grams of one mole of the compound. PubChem is a reliable source of chemical information from authoritative sources. Item Formula Molar Mass(g/mol) Mass of Sample (g) Moles of Sample (mol) Moles of Each. This document is not meant to be a substitute for a formal laboratory report. 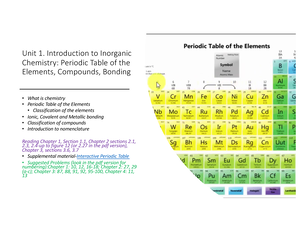
You can look up element names, symbols, atomic masses and more, or test your knowledge with a periodic table game. Molarity - Conversions and Mass Determination Hands-On Labs, Inc. A mole is the unit that measures the amount of a substance. Molarity And Mole Fraction - Get introduced to the concepts of molarity, molality, mole fraction, mass percent/weight percent and other related concepts. PubChem Periodic Table of Elements is an interactive tool that lets you explore the properties and trends of chemical elements. 
One thousand mers connected together would add up to a weight of 28,000 grams/mole and would have 6,000 atoms.Ī mole is the standard method in chemistry for communicating how much of a substance is present. We combine (react) many mers of ethylene together to form a polyethylene chain. Step 1: First, convert the mass of solute to moles using the molar mass of HCl (36.5 g/mol): 22.4 gHCl × 1 molHCl 36.5 gHCl 0.614mol HCl 22.4 g H C l × 1 m o l H C l 36.5 g H C l 0.614 m o l H C l. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) = 28. To calculate how to dilute a stock solution of known concentration. Please see the example above for how to use the Tocris molarity calculator to calculate the molarity of a solution. It is denoted by the unit M, and 1 M 1 mol/L. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms. Molar concentration (also known as molarity) is the amount of a solute (in moles) per unit volume of solution. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12.011 g/mol: Table 2.4.1 2.4. The chemical formula for an ethylene monomer is -(CH2-CH2)-. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole.In order to calculate the molecular weight of one water molecule, we add the contributions from each atom that is, 2(1) + 1(16) = 18 grams/mole. 
Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. The chemical formula for water is H2O, which means this molecule has 3 atoms: 2 of hydrogen (H) and 1 oxygen (O) atom. The volume and molarity of the solution are specified, so the amount (mol) of solute is easily computed as demonstrated in Example 4.3.3: M molsolute Lsolution.For example, if we wanted to know the mass of 0. 
A 355-mL soft drink sample contains 0.133 mol of sucrose (table sugar). The fact that a per relationship, ratio, exists between grams and moles implies that you can use dimensional analysis to interconvert between the two. Molarity is defined as the number of moles of solute in exactly 1 liter (1 L) of the solution: M mol solute L solution M mol solute L solution. The word per in this context implies a mathematical relationship between grams and mole. The atomic weights of aluminium and carbon are 26.98 and 12.01 respectively.Īs described in the previous section, molar mass is expressed as “grams per mole”. Three carbonate ions each containing three oxygen atoms gives a total of nine oxygens. To find the molar mass, we have to be careful to find the total number of atoms of each element. Aluminium carbonate, for example, contains aluminium, carbon, and oxygen. The procedure for more complex compounds is essentially the same. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
